GET INFUSED BY SGD PHARMA
GET INFUSED BY SGD PHARMA
One of the keys to SGD Pharma’s leadership position in the glass industry has always been its drive to continually innovate. Working closely with customers and researching the latest technologies, SGD Pharma seeks specific challenges to address through the research or application of new technology solutions. In essence, SGD Pharma connects products, processes and services to provide a unique and integrated packaging solution to meet ever-changing customer needs. SGD Pharma works closely with customers to solve their packaging challenges. We also focus on developing new innovations for the overall glass market.
SGD Pharma watches the glass market and its trends, to anticipate new needs and find solutions to address them. Our innovation is brought to life by our cross-functional teams, including R&D, Sales, Marketing, quality, regulatory, environment, health and safety, and operations, working together. We ensure new innovations area both technically viable and meet the exceptional quality and regulatory requirements our customer demand.
SGD Pharma works to solve tomorrow’s challenges today.
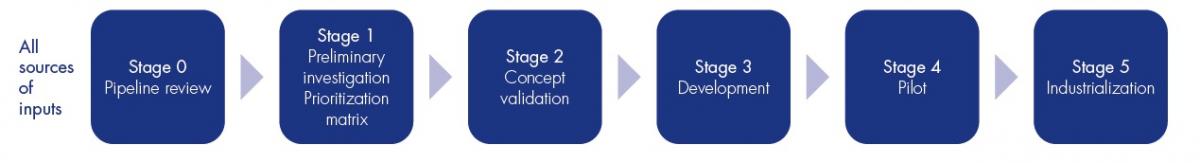
A clear process and a set of tools




































 Source: Webpackaging - 10 May 2022
Source: Webpackaging - 10 May 2022 GET INFUSED BY SGD PHARMA
GET INFUSED BY SGD PHARMA